A publication in Nature Neuroscience by Cyril Herry's team
Internally generated 4 hz oscillations synchronize prefrontal-amygdala neuronal circuits during fear behaviour.
Fear response to traumatic or threatening situations helps us evade or escape danger. At the same time fear response is learned in the form of association between stimulus or situation and the presence of a stressor (e.g. physical danger). This association is very powerful and leaves a memory trace that persists for years after a single experience, generating profound structural and functional changes in the brain that can potentially develop into post-traumatic stress and other anxiety-related disorders.
Read Cyril Herry's interview
Abstract
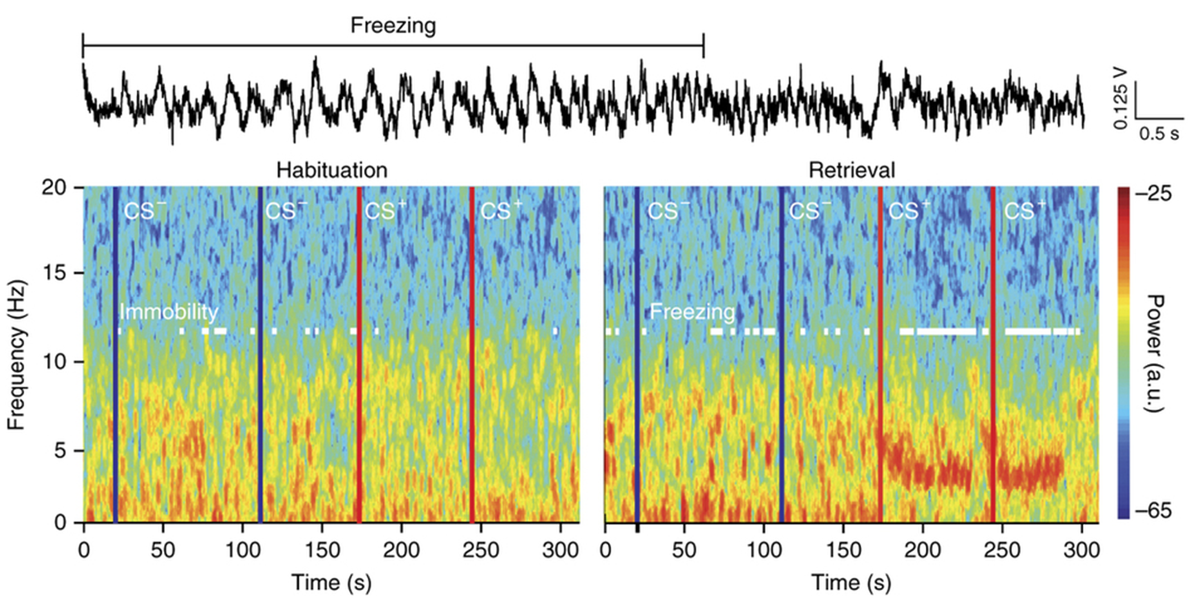
Fear expression relies on the coordinated activity of prefrontal and amygdala circuits, yet the mechanisms allowing long-range network synchronization during fear remain unknown. Using a combination of extracellular recordings, pharmacological and optogenetic manipulations, we found that freezing, a behavioral expression of fear, temporally coincided with the development of sustained, internally generated 4-Hz oscillations in prefrontal-amygdala circuits. 4-Hz oscillations predict freezing onset and offset and synchronize prefrontal-amygdala circuits. Optogenetic induction of prefrontal 4-Hz oscillations coordinates prefrontal-amygdala activity and elicits fear behavior. These results unravel a sustained oscillatory mechanism mediating prefrontal-amygdala coupling during fear behavior.
Nature Neuroscience, 2016 Apr;19(4):605-12
Authors: Karalis N, Dejean C, Chaudun F, Khoder S, Rozeske RR, Wurtz H, Bagur S, Benchenane K, Sirota A, Courtin J and Herry C

